藥物研發賦能解決方案
新藥伴隨診斷開發
新藥伴隨診斷開發
吉因加依託全鏈條解決方案,為藥企提供覆蓋“生物標誌物發現→方法學開發→臨床驗證與註冊申報→商業化”的一站式伴隨診斷服務。依託於現有的產品管線,我們深度參與多項伴隨診斷合作項目,覆蓋肺癌、腸癌、乳腺癌、膽管癌、黑色素瘤和前列腺癌等多個適應症。
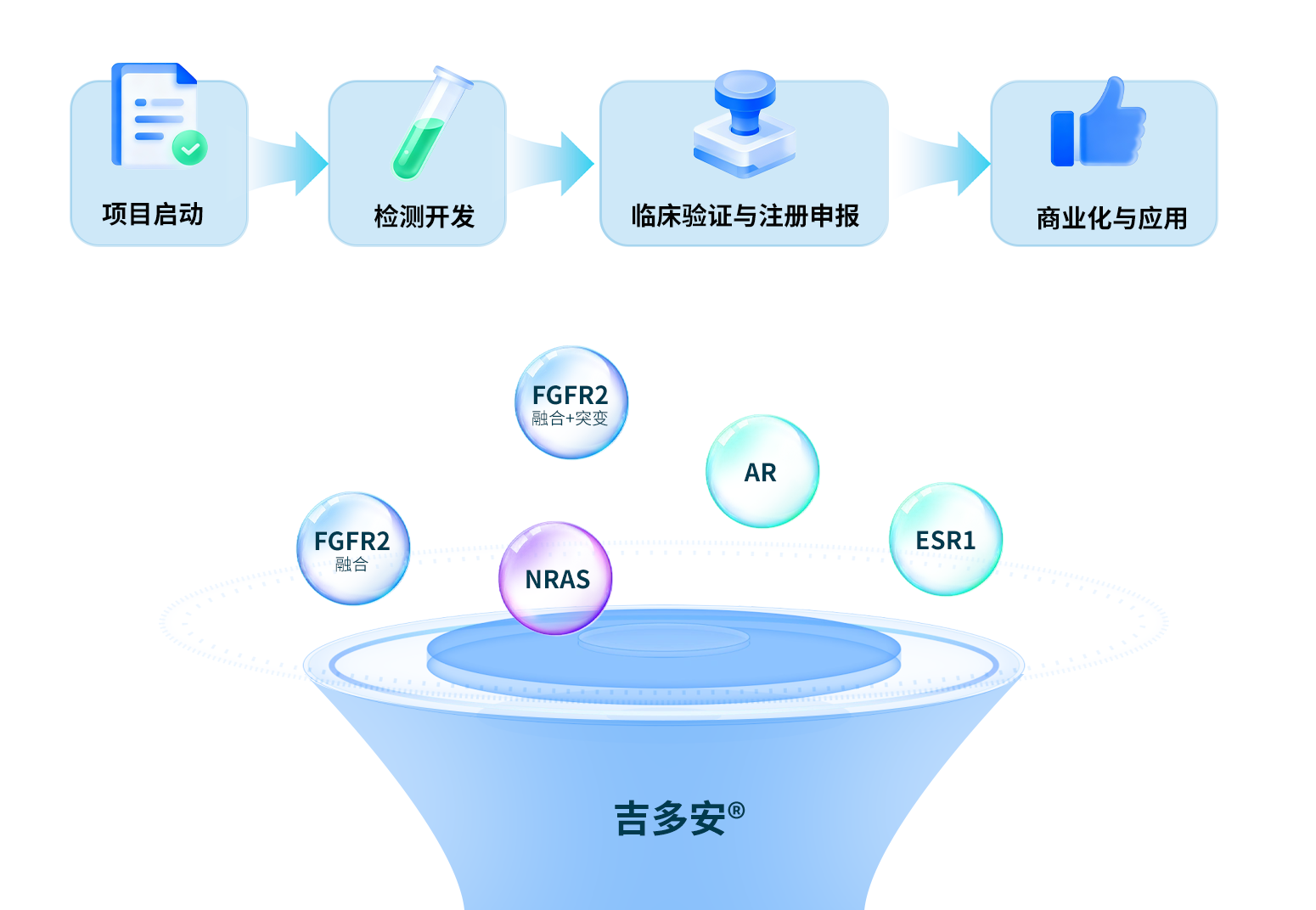
合作案例
吉因加作為佩米替尼(Pemigatinib)國內臨床試驗指定基因檢測中心實驗室,進行FGFR2重排患者的入組篩查。2022年4月6日,佩米替尼在國內成功上市,吉因加成為國內首個基於NGS技術助力FGFR2靶點新藥獲批的中心實驗室。
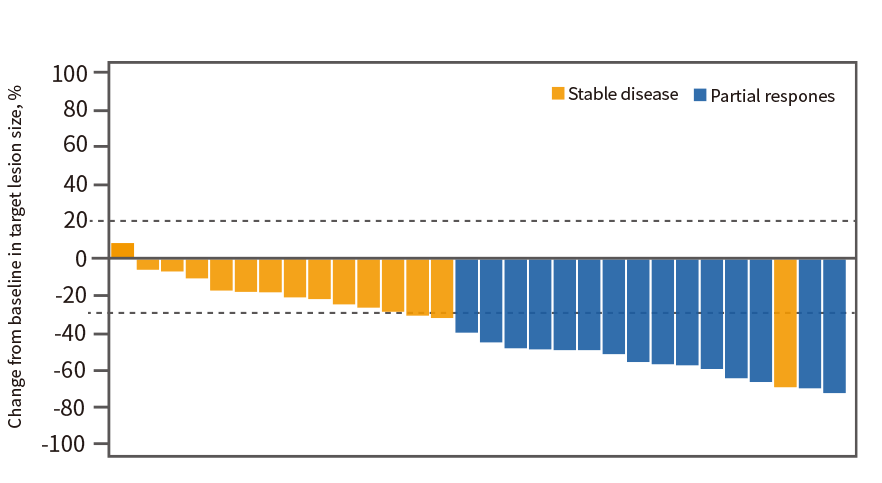
Endpoints and assessments
Patients in the phase II study were prescreened for FGFR2 status by next-generation sequencing (NGS) with the Malignant Neoplasms Multi-Gene Analysis Kit (Suzhou Geneplus Biomedical Engineering Co., Suzhou, China) at the central laboratory on archival formalin-fixed and paraffin-embedded tumor samples or fresh tumor samples; FGFR2 status reports based on DNA sequencing performed within 2 years could also be used; a relative abundance of variant allele of >5.3% was used to define FGFR2 fusions or rearrangements (the lower boundary of frequency detected by FMI central laboratory in FIGHT-202 study).
Cancer Medicine. 2023;12:4137-4146.

